Excited Cesium Atoms and the Radio Antenna
Fascinating read about an atomic receiver for AM and FM radio communication, able to operate from the C- to the Q-bands, which may someday bring quantum physics into the world of satcom receivers.
Here’s more from MIT Technology Review…
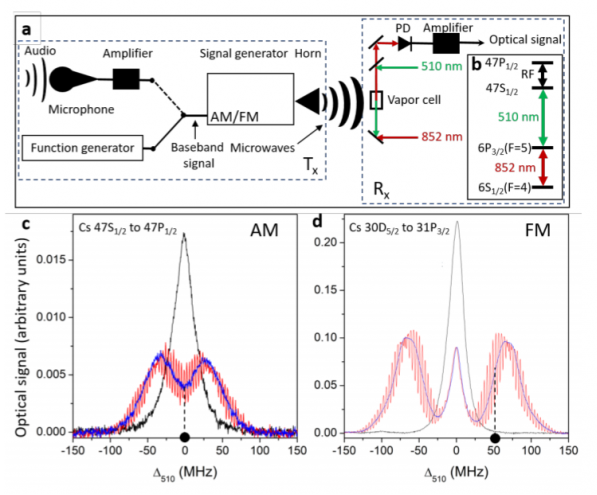
The secret sauce in the new device is Rydberg atoms. These are cesium atoms in which the outer electrons are so excited that they orbit the nucleus at great distance. At these distances, the electrons’ potential energy levels are extremely closely spaced, and this gives them special properties. Indeed, any small electric field can nudge them from one level to another.
Radio waves consist of alternating electric fields that readily interact with any Rydberg atoms they come across. This makes them potential sensors.
But how to detect this interaction? A gas made of Rydberg atoms has another property that turns out to be useful—it can be made transparent by a laser tuned to a specific frequency. This laser essentially saturates the gas’s ability to absorb light, allowing another laser beam to pass through it.
However, the critical frequency at which this happens depends crucially on the properties of the Rydberg atoms in the gas. When these atoms interact with radio waves, the critical frequency changes in response.
That’s the basis of the radio detection. Anderson and co create a gas of cesium atoms excited into Rydberg states. They then use a laser tuned to a specific frequency to make the gas transparent.
Finally, they shine a second laser through the gas and measure how much light is absorbed, to see how the transparency varies with ambient radio waves.
The signal from a simple light-sensitive photodiode then reveals the way the radio signals are frequency modulated or amplitude modulated.
And that’s it: an antenna consisting of a cloud of excited cesium atoms, zapped by laser light that flickers in time to any ambient radio waves. They call it atomic radio.
Very cool — and no electromagnetic interference!
Tags: antenna, atomic radio, c-band, ka-band, Ku-band, Q-band, quantum radio, radio, rydberg, s-band, X-band